Blood BHB and Cow Performance
6 min read
A DairyNZ study, within the Pillars of a new dairy system programme, investigated hyperketonaemia in grazing cows and determined that elevated blood beta-hydroxybutyrate (BHB) concentrations were not always associated with poor reproductive performance under a pasture-based diet.
At the start of lactation, dairy cows mobilise stored energy, protein, and mineral reserves from body fat, muscle, and bones to meet the high nutrient demands of milk production. This nutrient demand to support milk production often exceeds nutrient intake during early lactation and most cows experience negative energy balance, where body fat mobilisation releases fatty acids, which are converted to energy by the liver. When negative energy balance is severe, the liver can become overwhelmed, causing the production of ketone bodies, like beta-hydroxybutyrate (BHB).
Ketone bodies are an alternative energy source but when the cow's body can't use all the ketone bodies for energy, greater concentrations occur in the blood. Elevated blood BHB levels is called hyperketonaemia.
Notably, cows fed mainly on pasture have naturally higher blood BHB, and there are several factors that can influence blood BHB concentrations. In both pasture-based systems and housed systems abroad, moderately increased BHB concentrations aren't a standalone indicator of cow health and can simply be a sign of a high-producing cow, particularly after the first week of lactation.
Always consider other factors, including blood glucose and non-esterified fatty acid (NEFA) concentrations, body condition score, feeding levels, days post-calving and clinical symptoms. Routinely treating moderately hyperketonaemic cows with monopropylene glycol (MPG) drench isn't effective in pasture-based systems to improve economically important outcomes such as 6-week in-calf rate. A targeted approach, in consultation with a veterinarian, that prioritises treatment for cows with hyperketonaemia when clinical signs of disease are present is recommended.
All cows have some degree of negative energy balance during early lactation, where they lose body condition to provide energy for milk production. However, during severe negative energy balance, excessive amounts of body fat are mobilised, which the cow’s liver cannot convert to energy through the usual pathways.
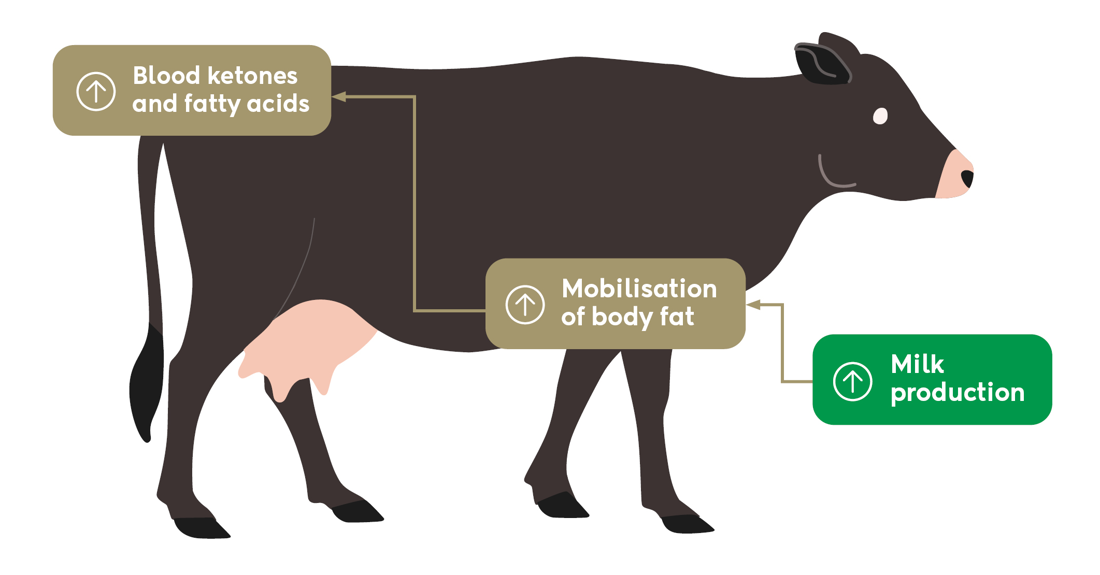
Instead, it produces ketone bodies, such as BHB, which the cow can use in small amounts as an alternative energy source. If ketone production becomes too high, the cow cannot use all the ketone bodies for energy and BHB concentrations increase in the blood. When this occurs, the cow may experience hyperketonaemia (an elevated concentration of BHB in blood).
Severe hyperketonaemia (blood BHB greater than or equal to 3.0 mmol/L) is associated with clinical ketosis and requires treatment. Severe hyperketonaemia affects about 6 - 18% of cows in the herd and can be accompanied by clinical signs such as lethargy, reduced feed intake, a characteristics sweet acetone odour on the breath, and rarely, abnormal excited behaviours such as excessive biting and licking.
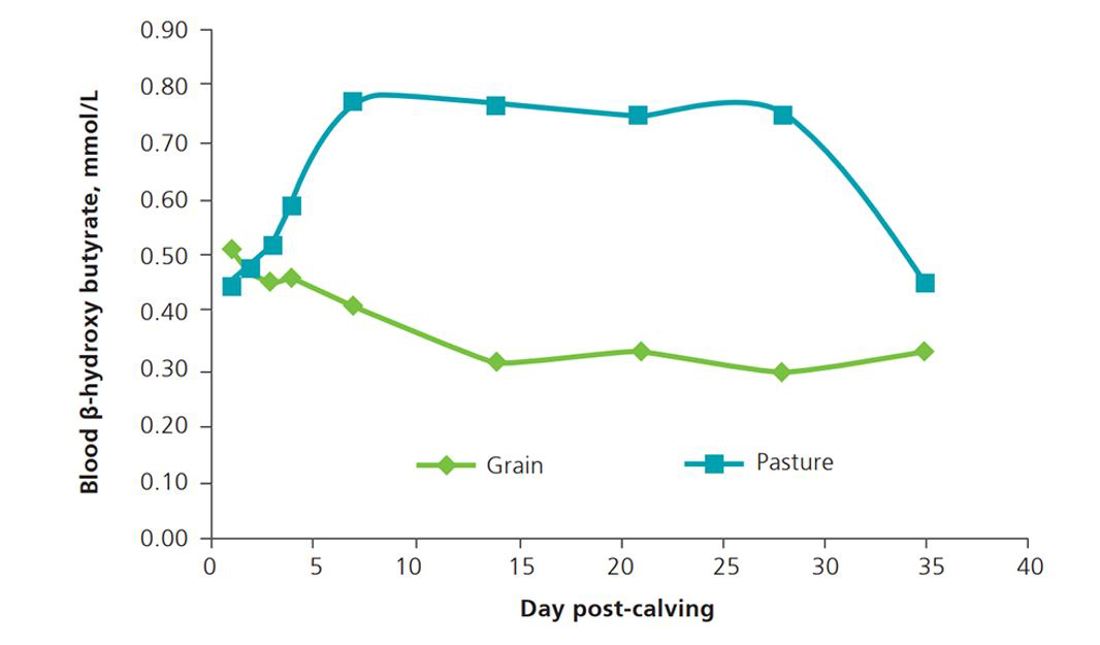
Figure 1. Blood beta-hydroxybutyrate concentrations in dairy cows fed the same energy as pasture (blue), or pasture plus grain (green). Milk energy output and BCS change were not different between groups (adapted from Roche et al., 2010).
Cows in early lactation are more likely to have hyperketonaemia with blood BHB concentrations between 1.2 and 2.9 mmol/L. This has been used to diagnose subclinical ketosis and described as a metabolic disease due to studies that report relationships between elevated blood BHB and adverse outcomes such as increased uterine infections and reduced reproductive performance in housed cows. However, these studies were not able to determine if elevated blood BHB concentrations directly caused undesirable outcomes or if it was due to a combination of other factors, which can contribute to inconsistencies between studies. Therefore, it has been suggested that hyperketonaemia (elevated blood BHB concentrations) is not a suitable standalone alone indicator of cow health challenges and in some cows, may simply occur due to the high energy demand experienced in early lactation.
Further, blood BHB is also affected by carbohydrate source in the diet. Blood BHB concentrations are naturally higher in cows fed predominantly pasture because they produce more butyrate in their rumens than those eating a diet high in starch such as grains or total mixed rations (Figure 1 - graph). Therefore, in cows grazing pasture, while hyperketonaemia can be a sign of health issues, it may also reflect a high-producing dairy cow and is not always associated with reduced cow performance.
Hyperketonaemia is common in pasture-based cows. A DairyNZ study in 2016 tested 980 cows in 3 herds for blood BHB three times per week for the first 5 weeks post-calving (Hendriks et al., 2025). The intensive sampling regime provided a very accurate pattern of hyperketonaemia, which although highly variable across the 3 herds studied, occurred most often during the first 7-10 days post-calving (Figure 2).
A positive test for hyperketonaemia was defined as a blood BHB test of 1.2 to 2.9 mmol/L. Overall, 76% of cows in these herds (across farms: min 65% and max 82%) had hyperketonaemia at least once during the first 5 weeks post-calving, whereas 11% of cows in these herds (across farms: min 6% and max 18%) had severe hyperketonaemia.
The percentage of cows with hyperketonaemia was slightly higher than in a 2010 study, where a subset of 565 cows from 15 herds in the Waikato and Canterbury regions were tested weekly for blood BHB concentrations (Compton et al., 2015). Overall, 67% of cows had hyperketonaemia (blood BHB greater than 1.2 mmol/L) at least once during the first 5 weeks post-calving; however, the degree and timing of hyperketonaemia varied significantly between herds (min 12%; max 100%).
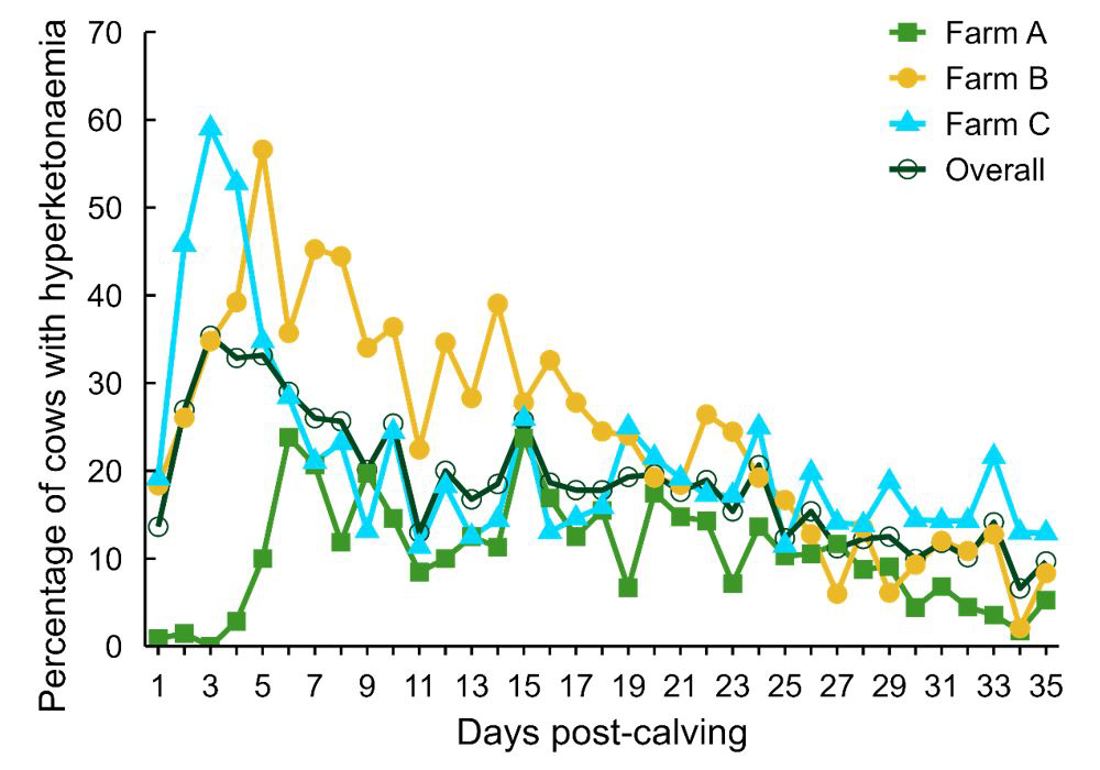
Figure 2. The daily % of cows that tested positive for moderate hyperketonaemia (blood BHB between 1.2 and 3.0 mmol/L) in 3 herds (Scott Farm, Waikato; Taranaki Agricultural Research Station (TARS), Taranaki; Lincoln University Dairy Farm (LUDF), Canterbury).
Many overseas studies conducted in housed cows have demonstrated relationships between elevated blood BHB concentrations (hyperketonaemia) and reduced reproduction and adverse health outcomes, but conflicting milk responses with both increased and decreased milk production reported. These studies explained the relationships between hyperketonaemia and a range of outcomes but could not determine if elevated blood BHB concentrations directly caused these effects. There is growing evidence that the BHB threshold concentrations used to define hyperketonaemia appear to be impacted by the outcome of interest and the timing of the blood BHB test relative to calving alongside various farm and system factors, which contribute to mixed results across studies.
In pasture-based cows these relationships are also unclear, with inconsistencies between studies. Similar to contrasting findings in housed cows, in the 2010 study, Compton et al. (2015) reported that cows with a BHB test over 1.8 mmol/L within the first 5 weeks post-calving had 15% lower milksolids production over the lactation, whereas in the DairyNZ study, cows with a blood BHB test over 1.2 mmol/L within the first 5 weeks post-calving produced about 4% more milk solids during the first 15 weeks in milk than those with a blood BHB test below 1.2 mmol/L.
Compton et al. (2015) reported that cows with blood BHB greater than 1.2 mmol/L within 5 days of calving were 2.5 times more likely to have a uterine infection at week 5 post-calving. Cows with a BHB test over 1.2 mmol/L at any stage during the first 5 weeks post-calving had a 7% lower 6-week In-Calf Rate. In contrast, the 2016 DairyNZ study did not determine any consistent relationships between hyperketonaemia (blood BHB of 1.2 to 2.9 mmol/L) during the first 5 weeks post-calving and subsequent uterine health and reproductive performance. There were no differences in the 6-week In-Calf rates overall and within each of the three farms (Figure 3).
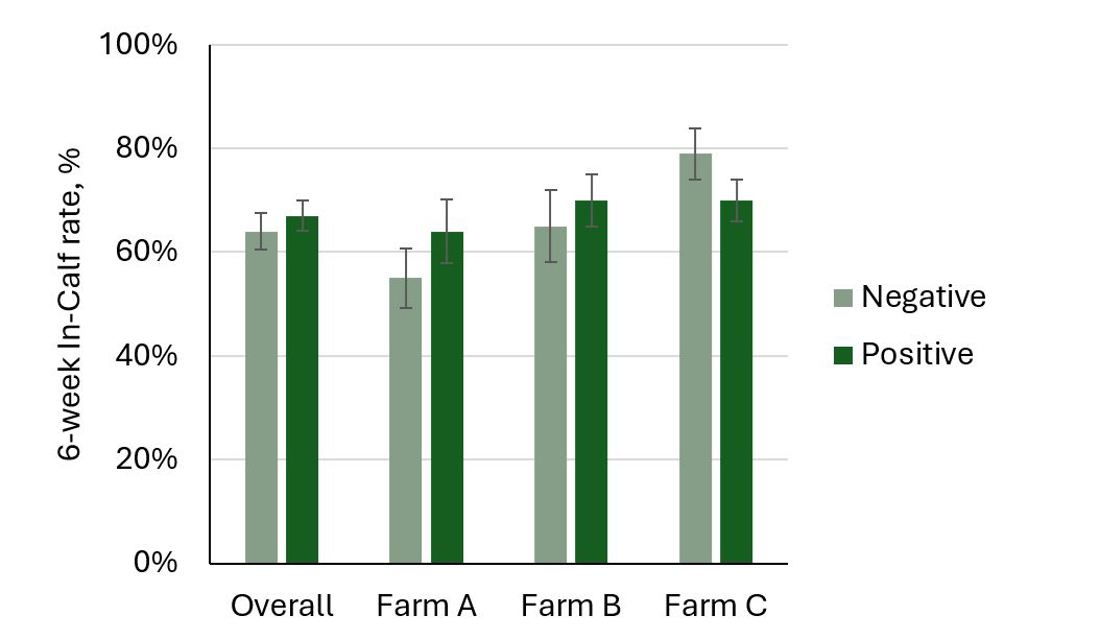
Figure 3. The 6-week In-Calf rates of cows that tested positive for hyperketonaemia (blood BHB of 1.2 to 2.9 mmol/L) at least once during the first 5 weeks post-calving compared with cows that only had negative tests (blood BHB below 1.2 mmol/L) in 3 herds (Farm A: Waikato; Farm B: Taranaki; Farm C: Canterbury).
Similar to Compton et al. (2015), the DairyNZ study also investigated the timing and severity of the blood BHB test on key outcomes and found that blood BHB concentrations over 1.2 to 1.6 mmol/L during the first week in milk did not impact 6 week In-Calf Rate, but when the blood BHB test was over 1.8 mmol/L, the 6 week In-Calf Rate was lower compared to cows with a blood BHB test of less than 1.8 mmol/L (Figure 4).
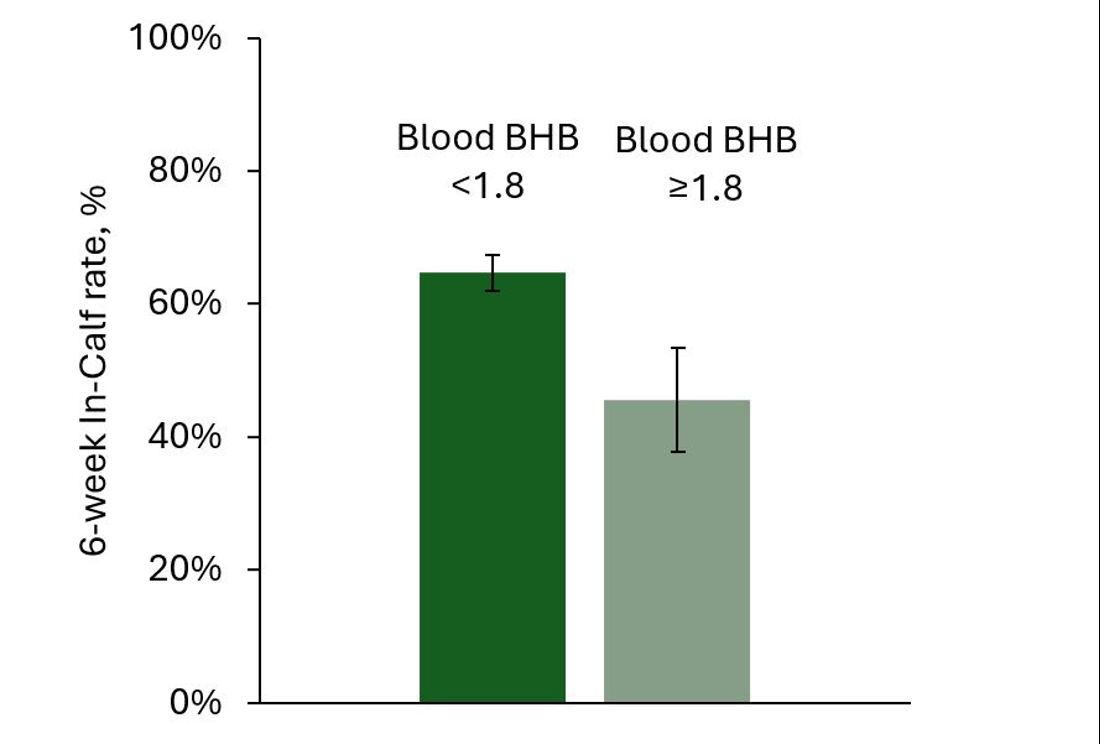
Figure 4. The 6-week In-Calf rate for grazing cows that had a blood BHB test of 1.8 to 2.9 mmol/L or a blood BHB test below 1.8 mmol/L within 7 days after calving.
Overall, the lack of consistent effects on key outcomes means that moderately elevated BHB concentrations, especially between 1.2 to 1.8 mmol/L, are not a suitable sole indicator of cow metabolic health status and poor outcomes in grazing cows. While higher BHB levels can be linked to negative effects, these results need to be interpreted alongside other clinical signs and factors. A combination of factors should be considered including blood fatty acids (non-esterified fatty acids) and glucose concentrations, BCS and feeding levels, clinical symptoms, and days post-calving.
Monopropylene glycol (MPG) is the active ingredient in energy drenches used to treat clinical ketosis. Overseas studies in housed systems also indicate that drenching of hyperketonaemic cows daily with MPG until blood BHB levels drop below 1.2 mmol/L can improve survival rates and reproductive performance (McArt et al. 2011; 2012). However, milk production benefits varied among herds indicating that the effectiveness of MPG drenching might depend on a range of different factors.
In the 2016 DairyNZ study, half of the cows with hyperketonaemia were drenched with MPG to determine if treatment was suitable in a New Zealand pasture-based system. Although treating hyperketonaemic cows with MPG temporarily reduced blood BHB concentrations and the likelihood they would subsequently test positive for severe hyperketonaemia (blood BHB greater than or equal to 3.0 mmol/L), there were no measurable improvements on reproductive performance overall, or within herds (Figure 5). Nor were there any marked improvements in milk production or uterine health outcomes.
These results indicate that using an MPG drench to treat cows with hyperketonaemia (without other symptoms of metabolic illness or clinical health issues) is not a suitable strategy to improve cow performance in pasture-based systems.
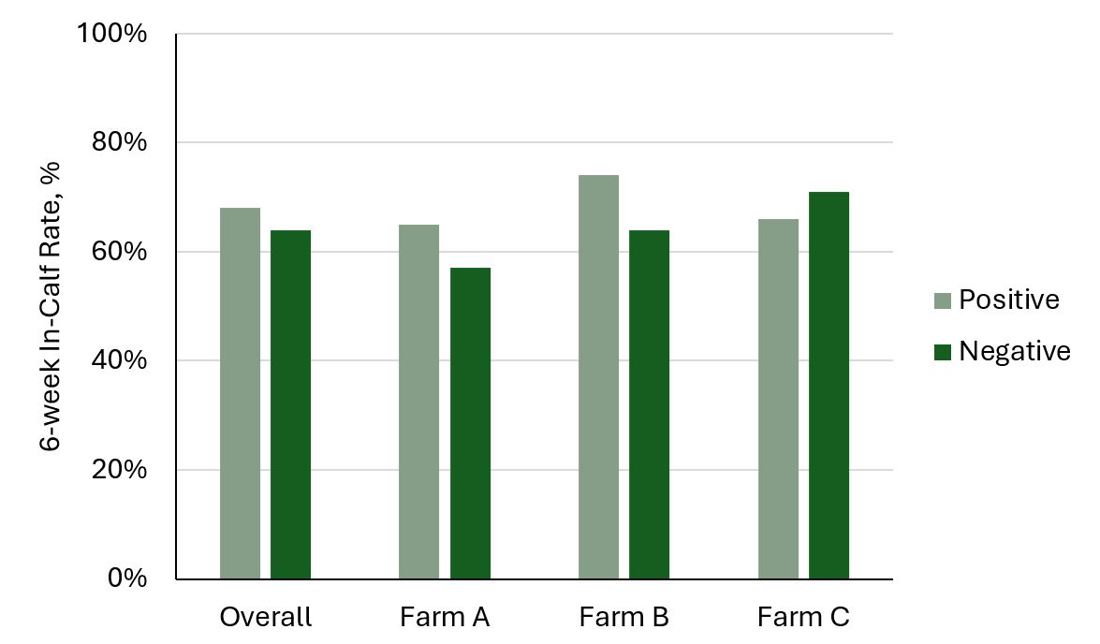
Figure 5. The effect of using monopropylene glycol (MPG) drench to treat cows diagnosed with hyperketonaemia (blood BHB of 1.2 to 2.9 mmol/L) on 6-week In-Calf rate in 3 herds (Farm A: Waikato; Farm B: Taranaki; Farm C: Canterbury).
Join Dr Claire Phyn, Dr Stacey Hendriks and Dr Jessica McArt in a session covering:
Compton et al. 2015. Subclinical ketosis in post-partum dairy cows fed a predominantly pasture-based diet; defining cut-points for diagnosis using concentrations of beta-hydroxybutyrate in blood and determining prevalence. New Zealand Veterinary Journal, 63:241-248.
Hendriks et al. 2025. Investigating the epidemiology of hyperketonemia in grazing dairy cows in early lactation: Incidence, prevalence, and time to resolution of hyperketonemia. Journal of Dairy Science, 108, 5257-5270.
Hendriks et al. 2025. The effect of monopropylene glycol on milk production, uterine health, and reproductive performance in cows diagnosed with hyperketonemia on 3 pasture-based dairy farms. Journal of Dairy Science, 108, 5271-5286.
Roche et al. 2010. Dietary structural to nonfibre carbohydrate concentration during the transition period in grazing dairy cows. Journal of Dairy Science, 93: 3671-3683.
McArt, J.A.A., D.V. Nydam, P.A. Ospina, and G.R. Oetzel. 2011. A field trial on the effect of propylene glycol on milk yield and resolution of ketosis in fresh cows diagnosed with subclinical ketosis. J. Dairy Sci. 94:6011–6020.
McArt, J.A.A., D.V. Nydam, and G.R. Oetzel. 2012. A field trial on the effect of propylene glycol on displaced abomasum, removal from herd, and reproduction in fresh cows diagnosed with subclinical ketosis. J. Dairy Sci. 95:2505–2512.
Now’s the perfect time to check in, plan, and set up for a strong season. We’ve pulled together smart tips and tools to help you stay ahead all winter long.
Whether you prefer to read, listen, or download handy guides, we’ve got you covered with trusted tools to support your journey every step of the way.
Put our proven strategies and seasonal tools to work. Boost production, support animal health and watch your profits hum.
Tools that are backed by science, shaped by farmers and made for this season.
That’s Summer Smarts.
Autumn Smarts brings together the research-backed tools that build resilience and boost performance.
The tools and insights you need to navigate the season with confidence. Backed by research and built for results – that’s Winter Smarts.